Still on eastern time, I was up early this morning, well before the crack of dawn. A quick walk outside revealed relatively mild temperatures, a subjective observation confirmed by the morning sounding from the Salt Lake City Airport, which showed a surface temperature of 38˚F and a freezing level near 8000 feet.
Based on temperature alone, one would expect rain on the valley floor and benches from a sounding like that, but a couple of hours later, it was snowing and accumulating on grassy surfaces.
Did a front go through, lowering the temperature, freezing level, and snow level? No. The lowering of temperature, freezing level, and snow level occurred due to sublimation, evaporation, and melting of precipitation. These processes are called diabatic effects by meteorologists.
To anticipate such effects, it is best not to use temperature, but instead wet-bulb temperature. Wet-bulb temperature is the temperature that air will cool too if you evaporate water into it. When the relative humidity is 100%, the wet-bulb temperature equals the temperature, but when the relative humidity is less than 100%, the wet-bulb temperature is lower than the temperature.
Most Utahns have first-hand experience with the cooling effects of evaporation. A swamp cooler, for example, can potentially cool the air to the wet-bulb temperature. When the relative humidity is low, a swamp cooler can lower the air temperature significantly. When the relative humidity is high, however, the cooling effect is limited.
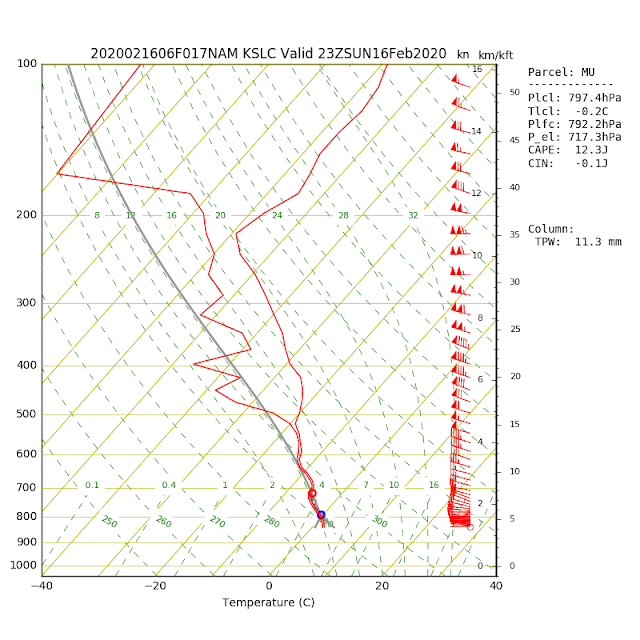
Today's sounding was primed for substantial cooling. If you look at it, there is a large difference between the air temperature (red line) and the dew point (green line) below 700 mb (10,000 ft). There is a thin blue line between the temperature and dew point lines that is the profile of wet-bulb temperature. Note that the wet-bulb temperature is below 0˚C above 850 mb (about 5,000 ft). Thus, if you can precipitate into that layer, the evaporation of liquid water and the sublimation of snow can potentially drop the freezing level to about 5000 feet, with the snow level just a bit lower than that.
This is what happened. This morning, when precipitation increased, diabatic processes led to a decrease in temperature and a dramatic lowering of the snow level.
A look at surface observations shows, however, that we haven't quite cooled to the wet-bulb temperature. Below is the temperature trace at the University of Utah. At about 7 AM (far right side of the graph), the temperature (red line) decreased abruptly, the dewpoint (green line) rose abruptly, and the relative humidity (blue line) increased abruptly). This is consistent with cooling and moistening through evaporation and sublimation, with temperature and dew point converging to the wet bulb temperature.
However, they don't quite meet. The relative humidity ends up at about 90%. Thus we are close but not quite at the dew point temperature.
There is one other effect that I've swept under the rug here and that is melting. With temperatures above 0˚C, the flakes falling at my house are also melting. This also takes energy out of the atmosphere, leading to a cooling effect. It is probably playing some role as well.
In situations like today's, precisely predicting snow level and, for the valley and benches whether or not the precipitation will be snow or rain, can be challenging. A look at NAM forecast soundings shows temperatures at all levels below 0˚C at 1500 UTC (0800 MST). Note, however, that the NAM terrain is higher than the real-world terrain, so in reality, we're just above 0˚C on the benches and in the valley.
Later today, however, temperatures in the lower part of the sounding have climbed to > 0˚C as warmer air moves in from the west and we get a little daytime surface heating. This yields a higher freezing level (near 800 mb or about 6500 feet).
This will probably mean snow levels will rise today, resulting in the bench snow turning to rain later this morning or this afternoon.





Jim, that was one of your most enlightening posts! Diabatic fascination!
ReplyDeleteditto
ReplyDelete